7. Drug Updates
Oral semaglutide reduces MACE risk by 14% in type 2 diabetes – Reveals SOUL study
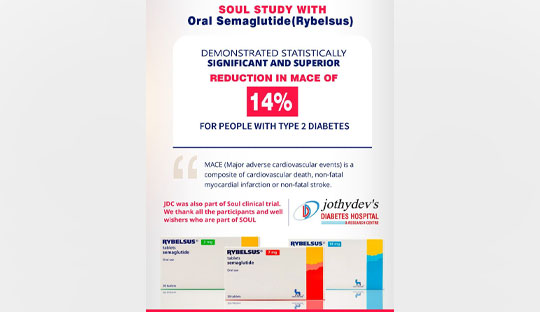
The SOUL trial, initiated in 2019, was a multicenter, international, randomized, double-blind, parallel-group, placebo-controlled phase 3 study. Its primary goal was to assess the impact of oral semaglutide on cardiovascular outcomes in individuals with type 2 diabetes and established CVD and/or CKD. The trial included 9650 people with type 2 diabetes coupled with either cardiovascular disease or chronic kidney disease and were randomly assigned to receive either oral semaglutide or a placebo, with both groups receiving standard care. The composite measure of cardiovascular death, non-fatal myocardial infarction, or non-fatal stroke was referred to as MACE in this context. SOUL results revealed that oral semaglutide significantly reduces MACE risk by 14% in people with type 2 diabetes. The observed decrease was a result of each component of this composite measure, highlighting the efficacy of oral semaglutide Safety and tolerability of oral semaglutide remained consistent with prior trials, indicating it is well-tolerated by patients.
For enquiries info@jothydev.net.
Please visit: jothydev.net | research.jothydev.com | diabscreenkerala.net | jothydev.com/newsletter




