2. Can Stem Cells Train the Immune System to Fight Type 1 Diabetes?
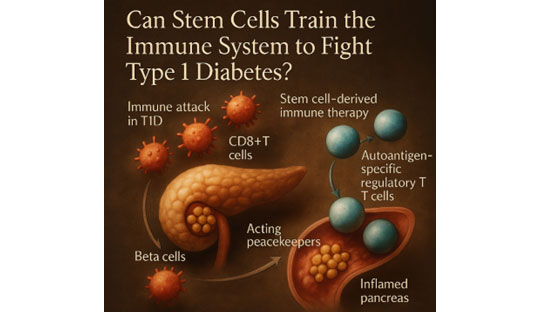
A new frontier in T1D treatment: Using stem cell-derived regulatory T cells to calm immune attacks
Type 1 diabetes (T1D) occurs when the immune system mistakenly destroys insulin-producing beta cells in the pancreas. But what if we could retrain the immune system to stop the attack? In a groundbreaking study, scientists used induced pluripotent stem cells (iPSCs) to create a special type of immune cell—autoantigen-specific regulatory T cells (iPSC-Tregs)—engineered to target and suppress autoimmune responses in T1D.
Background:
Type 1 diabetes (T1D) is an autoimmune disorder where the body’s own immune system—particularly CD8+ T cells—destroys insulin-producing β cells in the pancreas. While insulin therapy is the mainstay of treatment, it does not address the root immune dysfunction. Traditional β cell replacement therapies often fail due to ongoing immune attack. Regulatory T cells (Tregs) have emerged as a promising tool to suppress autoimmunity, but challenges in expanding sufficient Treg populations have limited clinical application—until now.
What Did the Study Show?
Using a diabetic mouse model, researchers found that:
- Adoptive transfer of iPSC-Tregs significantly prevented destruction of pancreatic beta cells.
- These cells migrated directly to the inflamed pancreas, acting like peacekeepers.
- Once there, they reduced the levels of ICAM-1, a molecule that helps harmful immune cells stick to inflamed tissue.
- This, in turn, blocked CD8+ T cells (the main attackers in T1D) from entering the pancreas.
- They also reduced pro-inflammatory IFN-γ, a chemical signal that ramps up the immune attack.
Why This Matters
This study suggests that stem cell–derived immune therapy could be a powerful way to stop or slow the autoimmune process in type 1 diabetes—without broadly suppressing the immune system.
Instead of just managing blood sugar levels, future therapies might actually target the root cause of T1D: immune misfiring.
Key Scientific Insights
-
Generation & Identity:
Autoantigen-specific iPSC-Tregs were successfully derived from iPSCs. These cells expressed hallmark Treg markers—FoxP3, CD25, and produced IL-10—demonstrating strong similarity to natural regulatory T cells (nTregs). -
Disease Prevention:
Adoptive transfer of these iPSC-Tregs into diabetic mice prevented β-cell destruction, maintained normal blood glucose levels, and reduced diabetes incidence significantly. -
Mechanism of Action:
The iPSC-Tregs suppressed autoimmune responses by reducing ICAM-1 expression in the pancreas, which limited the migration of pathogenic CD8+ T cells and decreased pro-inflammatory IFN-γ production. -
Tissue Targeting & Antigen Specificity:
These Tregs specifically homed to the inflamed pancreatic tissue, where they were most active. Their autoantigen specificity enabled precise suppression of harmful immune responses without general immunosuppression. -
Cytokine-Mediated Immune Regulation:
In the pancreas, iPSC-Tregs produced significantly higher levels of IL-10 and TGF-β, reinforcing their local immune-regulatory effect and helping protect insulin-secreting islets from immune attack.
GEMS Takeaway
Science is getting closer to teaching the immune system how to protect rather than attack. This study adds hope that cell-based therapies could one day prevent or even reverse type 1 diabetes by creating precision immune modulators from a person’s own cells.
For enquiries info@jothydev.net.
Please visit: jothydev.net | research.jothydev.com | diabscreenkerala.net | jothydev.com/newsletter




