7. Device and Drug Updates
GEMS Device Update: The First OTC CGM App for Weight Management Gets FDA Clearance
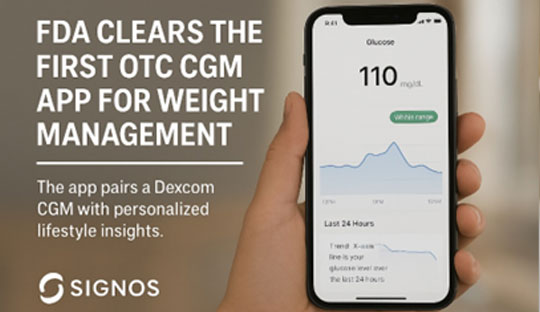
FDA Clears a New Path to Personal Weight Control
For the first time, the FDA has cleared an over-the-counter (OTC) continuous glucose monitoring system designed specifically for weight management, not diabetes. Developed by Signos, this innovative system pairs Dexcom’s already OTC-cleared Stelo CGM sensor with a personalized smartphone app—marking a groundbreaking expansion of CGM technology into wellness and metabolism tracking.
How It Works
Using the Stelo CGM, the Signos platform monitors your glucose levels in real time and translates daily fluctuations into tailored lifestyle insights. The app helps users understand how food, stress, sleep, and activity affect metabolism—then makes practical suggestions to guide healthier habits. It also integrates nutrition counseling—courtesy of partner startup Nourish—for added dietary support.
Accessibility & Cost
The system is available via a quarter-long subscription that includes six CGM sensors for $139 per month. As a wellness tool, it clarifies that medical decisions based on app output should only be made in consultation with a healthcare provider.
Why This Matters for People with or at Risk for Diabetes
CGMs were once restricted to diabetes management, but this FDA clearance opens doors for broader metabolic awareness—by helping users adapt and optimize lifestyle behaviors based on individual glucose responses. Such real-time feedback can be especially valuable alongside weight loss medications (like GLP-1s) or post-surgery strategies.
GEMS Takeaway
With the clearance of Signos’ OTC CGM app, people now have a science-backed, tech-driven tool to better understand their bodies and make informed lifestyle changes. It is not a medical device—yet it empowers weight and metabolism self-management with a level of personalization rarely seen before. Always remember: use these insights wisely and in tandem with professional healthcare advice.
Wegovy® Receives FDA Approval for Treatment of Noncirrhotic MASH with Moderate to Advanced Fibrosis

Overview
The U.S. Food and Drug Administration (FDA) has granted accelerated approval to Wegovy® (semaglutide injection 2.4 mg) for the treatment of adults with metabolic dysfunction-associated steatohepatitis (MASH) and moderate to advanced liver fibrosis (stage F2–F3) without cirrhosis. Approved on August 15, 2025, this represents a significant step forward in liver disease therapeutics, as Wegovy becomes the first and only GLP 1 receptor agonist (GLP 1 RA) approved for this indication. Treatment must be used in conjunction with a reduced-calorie diet and increased physical activity, underscoring the importance of lifestyle modification alongside pharmacotherapy. The approval is based on data from Part 1 of the Phase 3 ESSENCE trial, which demonstrated significant histological improvements over 72 weeks.
Why This Matters
MASH is a serious, progressive liver disease often detected late, affecting roughly 1 in 20 adults in the U.S. Without intervention, it can lead to cirrhosis, liver failure, or cancer. This approval addresses a major unmet medical need.
Key Findings:
The ESSENCE trial (Part 1) enrolled adults with biopsy-confirmed MASH and moderate to advanced fibrosis and evaluated the effects of once-weekly Wegovy 2.4 mg over a 72-week period. The findings were compelling: 63% of patients treated with Wegovy achieved resolution of steatohepatitis without worsening of fibrosis, compared to approximately 34% in the placebo group. Furthermore, 37% experienced improvement in fibrosis without worsening of steatohepatitis, versus 22% in the placebo arm. Importantly, 33% of Wegovy-treated patients achieved both outcomes (both inflammation resolution and fibrosis improvement) compared to around 16% in the control group. The trial also reported strong medication adherence, with 83.5% of patients maintaining the target 2.4 mg dose through Week 72. These results support Wegovy’s efficacy in targeting both inflammation and fibrosis, the two key pathological hallmarks of MASH. The ongoing Part 2 of the ESSENCE trial will assess whether Wegovy leads to a reduction in long-term liver-related clinical events, including progression to cirrhosis, over a 240-week period.
MASH, formerly known as NASH (non-alcoholic steatohepatitis), is a serious liver condition with rising global prevalence. In the U.S., it is estimated that around 5% of adults are affected. Alarmingly, one in three individuals with overweight or obesity is believed to have MASH worldwide. If untreated, the condition may progress to cirrhosis, hepatocellular carcinoma, or lead to the need for liver transplantation. Despite this disease burden, until now there were no FDA-approved therapies specifically indicated for treating MASH with fibrosis.
Wegovy’s latest approval adds to its growing list of indications. Originally approved in 2021 for chronic weight management in adults, it received an extension in 2022 for treating obesity in adolescents aged 12 years and older. In 2024, the FDA approved Wegovy for cardiovascular risk reduction in adults with obesity or overweight and established heart disease. With the current approval for MASH, Wegovy now holds four distinct FDA indications. Additionally, regulatory submissions for this indication have been made in the European Union (February 2025) and Japan (May 2025), indicating Novo Nordisk’s intention to expand global access.
GEMS Take Away
Wegovy® (semaglutide 2.4 mg) has emerged as a transformative therapy for metabolic liver disease, being the first GLP-1 RA approved to treat MASH with fibrosis. The robust histological improvements demonstrated in the ESSENCE trial—particularly the resolution of steatohepatitis and reversal of fibrosis—highlight its potential to change the treatment paradigm for this high-burden condition. High adherence rates reinforce its real-world viability. As MASH frequently coexists with obesity, diabetes, and cardiovascular disease, Wegovy’s expanding utility across multiple metabolic conditions makes it a versatile and strategic therapeutic option.
For clinicians, this approval represents a major development in hepatometabolic care. It not only provides a pharmacological option for a previously untreatable condition but also reinforces the importance of comprehensive lifestyle management. Wegovy® now offers hope beyond weight management—it targets the underlying liver damage in adults with MASH and significant scarring. As the first GLP 1 therapy approved for this use, it could transform care for thousands silently at risk. And while it works best alongside healthier lifestyle choices, it's a promising therapeutic leap forward in liver health.
For enquiries info@jothydev.net.
Please visit: jothydev.net | research.jothydev.com | diabscreenkerala.net | jothydev.com/newsletter




