7. Drug Updates
Baxdrostat: A Novel Oral Option for Hard-to-Control Hypertension
What’s New:
Baxdrostat is an investigational once-daily oral drug showing promise as a new treatment for uncontrolled and resistant hypertension, a condition in which blood pressure remains elevated despite multiple medications. This novel approach could be particularly relevant for people with diabetes, who face an elevated risk of cardiovascular and renal complications when blood pressure is not well controlled.
Mechanism of action:
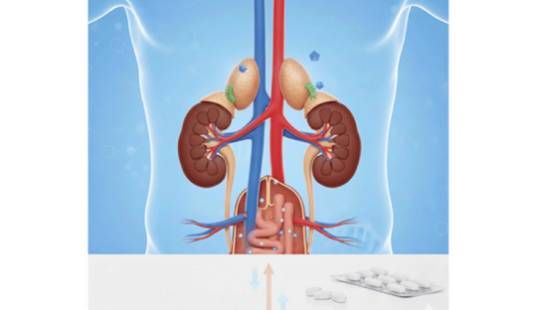
Unlike traditional antihypertensive agents (such as ACE inhibitors, ARBs, diuretics or calcium channel blockers) that primarily block hormone effects or vascular resistance, baxdrostat targets aldosterone production itself. Aldosterone, a hormone produced by the adrenal glands, promotes salt and water retention that increases blood volume and raises blood pressure. By selectively inhibiting aldosterone synthase, baxdrostat reduces aldosterone levels, helping the kidneys excrete excess sodium and water and thereby lowering blood pressure.
Clinical evidence:
Results from the Phase III BaxHTN trial have been encouraging. In a global study involving patients with uncontrolled or resistant hypertension already on standard therapy, once-daily baxdrostat (at 1 mg or 2 mg doses) produced significant reductions in systolic blood pressure compared with placebo at 12 weeks, with placebo-adjusted decreases in seated systolic blood pressure of approximately 8–10 mmHg. A substantial proportion of patients treated with baxdrostat also achieved target blood pressure levels (<130 mmHg) versus placebo.
The drug was generally well tolerated, with no unexpected safety findings and low rates of significant hyperkalaemia reported. 24-hour ambulatory blood pressure monitoring in exploratory analyses suggested durable and sustained blood pressure lowering including during high-risk early morning hours compared with placebo.
Regulatory progress:
AstraZeneca has submitted a New Drug Application (NDA) for baxdrostat to the U.S. Food and Drug Administration, and the agency has granted Priority Review. A decision is anticipated in 2026. If approved, baxdrostat would represent a first-in-class aldosterone synthase inhibitor for patients with uncontrolled hypertension.
Clinical implication:
Resistant hypertension remains a major clinical challenge: many patients with diabetes fail to reach target blood pressure despite combinations of established drugs. By directly targeting aldosterone synthesis, a key hormonal driver of blood pressure and fluid balance, baxdrostat promises an alternative, complementary mechanism that may benefit those whose hypertension persists despite conventional therapy.
GEMS Takeaway:
Baxdrostat could become a valuable new oral option for treatment-resistant and uncontrolled hypertension by suppressing aldosterone production, a mechanism distinct from existing antihypertensives. With robust Phase III data showing meaningful blood pressure reductions and a favorable tolerability profile, ongoing regulatory review may soon expand clinicians’ tools for managing high blood pressure in high-risk populations, including people living with diabetes.
For enquiries info@jothydev.net.
Please visit: jothydev.net | research.jothydev.com | diabscreenkerala.net | jothydev.com/newsletter




