7. Device and Drug Updates
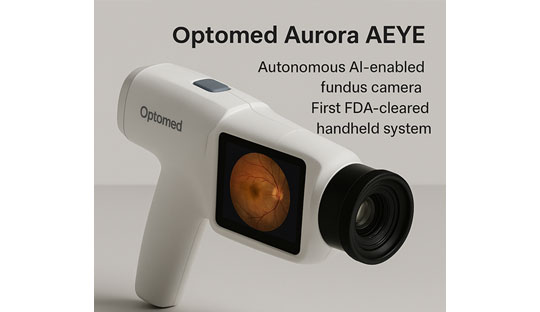
Optomed Aurora AEYE: A Game Changer in Diabetic Retinopathy Screening
Why This Matters for Diabetes Care?
The Optomed Aurora AEYE is the first FDA cleared handheld AI-enabled fundus camera designed specifically for autonomous detection of referable diabetic retinopathy (DR). This device represents a major leap in screening technology: portable, fast, accurate, and suitable for non-ophthalmic settings, meaning earlier detection and potential prevention of vision loss in people living with diabetes.
Key Features
- Autonomous AI diagnostic capability: Using the embedded AEYE-DS software, the Aurora AEYE can analyse a single image per eye and deliver a screening result in under 60 seconds, no clinician needed for the interpretation.
- Strong clinical performance: In large-scale phase III clinical studies, the AI achieved sensitivities of ~92–93% and specificities of ~89–94%, with over 99% imageability (i.e., a valid result obtained) using non-mydriatic (undilated) imaging.
- Handheld and portable design: The compact form factor allows use in primary care clinics, endocrinology practices, screening camps, and remote settings rather than being confined to specialist eye clinics.
- Streamlined workflow: One image per eye → automated AI verdict → result displayed on-device. This simplicity promotes high screening throughput and can integrate into diabetes-care pathways.
- Regulatory clearance & global readiness: FDA cleared in the U.S., CE-marked in Europe, and present in key global markets.
Implications for Diabetes-Care Teams
- Closing the screening gap: Many people with diabetes miss regular retinal screening due to access, cost or specialist availability. A handheld AI camera empowers non-eye care providers (e.g., endocrinologists, diabetologists, general practitioners) to screen on-site, improving early detection.
- Timely referral and intervention: Rapid, on-the-spot results mean that patients flagged as “referable” can be fast-tracked to ophthalmology, reducing the window of unnoticed progression.
- Resource optimisation: Given its high specificity, fewer false positives translate into less unnecessary specialist load, and high sensitivity ensures fewer missed cases.
- Scalable in low-resource / outreach settings: The portability and simplicity make this camera suited for rural or resource-limited regions, potentially transforming screening coverage in countries like India.
- Patient empowerment & engagement: Having screening done during routine diabetes visits may boost uptake as patients experience the convenience and immediacy of results.
GEMS Takeaway
The Optomed Aurora AEYE heralds a new era in diabetic retinopathy screening by enabling accurate, rapid, autonomous detection of sight-threatening changes in a handheld format. For diabetes care teams, this offers a practical tool to bridge the gap between endocrinology and ophthalmology, embed retinal screening into routine visits, and ultimately protect vision in people living with diabetes.

Rybelsus (oral semaglutide) - A New Milestone in Cardiovascular Prevention for People with Type 2 Diabetes
Rybelsus (semaglutide) is the first oral GLP-1 receptor agonist approved to help adults with type 2 diabetes improve blood glucose control and, in some cases, reduce cardiovascular risk. It works by mimicking the natural GLP-1 hormone, enhancing insulin release when blood sugar is high, lowering glucagon levels, and slowing stomach emptying to promote fullness and reduce appetite.
The recent update to the indication for Rybelsus marks a significant advance in cardiometabolic care. Previously approved in 2019 for glycaemic control in adults with type 2 diabetes (T2D), Rybelsus has now become the first oral GLP-1 receptor agonist approved for reducing the risk of major adverse cardiovascular events (MACE) including cardiovascular death, non-fatal myocardial infarction, and non-fatal stroke in adults with T2D who are at high cardiovascular risk.
Key Findings from the SOUL Trial
- The approval is based on results from the SOUL trial, which included over 9,600 adults with T2D and cardiovascular or kidney disease.
- Rybelsus 14 mg reduced the risk of MACE by 14% compared with placebo (HR 0.86; 95% CI 0.77–0.96; p = 0.006).
- The benefit was seen in both primary and secondary prevention, including patients without prior cardiovascular events.
- Safety profile: Rybelsus was generally well tolerated, with slightly fewer serious adverse events vs placebo (47.9% vs 50.3%). Gastrointestinal side effects such as nausea and vomiting were more common but consistent with the known class profile.
What This Means for Practice
For clinicians managing T2D patients, especially those with established cardiovascular disease or chronic kidney disease (or both), the expanded Rybelsus indication offers a once-daily oral therapy that goes beyond glycaemic control, potentially reducing cardiovascular risk. This is especially relevant in settings where injectable therapies are less feasible or accepted. It underscores the importance of integrated cardiometabolic care, treating blood glucose, cardiovascular risk and kidney health in tandem.
Practical Considerations
- Indication: Adults with T2D and established cardiovascular disease or at high cardiovascular risk.
- Dosage: 7 mg or 14 mg oral tablet once daily.
- Administration: Take in the morning on an empty stomach with up to 120 mL (4 oz) of plain water. Wait at least 30 minutes before eating, drinking (other than water), or taking other medicines.
- Missed dose: Skip the missed tablet and take the next dose as usual, do not double up.
- Adverse effects: Nausea, vomiting, and diarrhea are the most common; these are usually mild and transient. Gradual dose escalation can improve tolerance.
- Contraindications: Personal or family history of medullary thyroid carcinoma or multiple endocrine neoplasia type 2 (MEN-2); use with caution in those with a history of pancreatitis.
- Lifestyle advice: Maintain a balanced diet, regular exercise, and limit alcohol intake to minimize fluctuations in blood glucose.
GEMS Takeaway
The expanded indication for Rybelsus significantly elevates its role in cardiometabolic care: for adults with T2D and elevated cardiovascular risk, it represents a dual-benefit therapy, targeting both glycaemic control and cardiovascular protection delivered orally.
In practice, this underscores the evolving paradigm: glucose control alone is not enough, we must aim for cardiovascular and kidney protection. Rybelsus provides a new tool to help achieve that broader goal.
For enquiries info@jothydev.net.
Please visit: jothydev.net | research.jothydev.com | diabscreenkerala.net | jothydev.com/newsletter




