4. CRISPR-Engineered Islet Cells Evade Immune Rejection and Restore Insulin Secretion in First-in-Human Study
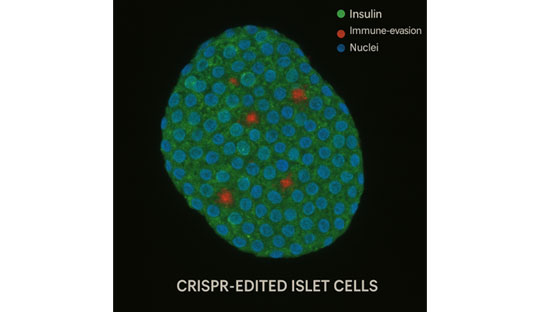
A major advance in immune-evasive, cell-based therapy for Type 1 Diabetes
Background: Overcoming Immunological Barriers in Islet Cell Replacement
Islet transplantation has long been recognized as a promising therapy for Type 1 Diabetes Mellitus (T1DM). However, the approach is limited by the requirement for lifelong systemic immunosuppression to prevent graft rejection. This not only increases the risk of infection and malignancy but also limits clinical scalability. A durable solution requires immune evasive cells that can function physiologically without provoking host immune responses.
Study Overview: In Vivo Functionality of CRISPR-Edited Human Islets
In a landmark first-in-human trial, researchers have implanted CRISPR-Cas9 gene-edited pancreatic islet cells into a person with T1DM. These cells were engineered to:
- Knockout two major immune-regulatory genes that present alloantigens, thereby minimizing adaptive immune recognition.
- Introduce the CD47 gene, which acts as an innate immune checkpoint by delivering a “don’t eat me” signal to natural killer (NK) cells and macrophages.
The dual-editing strategy aims to attenuate both innate and adaptive immune-mediated destruction of transplanted islets, creating a state of functional immune evasion.
Key Findings
The edited islet cells successfully secreted insulin in vivo and persisted without immunosuppressive therapy. Notably:
- The graft demonstrated glucose-responsive insulin secretion, suggesting restoration of β-cell function.
- There were no signs of immune rejection, validating the effectiveness of the immune-editing strategy.
- This marks the first clinical demonstration of immune-stealth allogeneic islet cells functioning in a human host.
Translational Significance
If replicated at scale, this strategy offers several transformative implications:
- Enables the use of allogeneic or stem-cell–derived islets without the need for HLA matching or immunosuppression.
- Provides a blueprint for universal donor cells for diabetes and potentially other autoimmune or alloimmune indications.
- May improve the long-term viability and function of implanted β-cell sources in regenerative medicine.
Future directions include optimizing gene-editing platforms, ensuring genomic stability of edited cell lines, and scaling up through pluripotent stem cell derived islets, which offer an unlimited cell source for therapeutic use.
GEMS Perspective: Convergence of Gene Editing and Endocrine Regeneration
This study represents a critical convergence of gene-editing, immunology, and β-cell biology. By successfully integrating immune-evasion strategies into functional insulin-producing cells, researchers have taken a decisive step toward curative, immune-independent treatment for T1DM.
The long-term impact of this approach could redefine the paradigm of diabetes care—shifting from external insulin delivery to endogenous insulin restoration via engineered cell therapy. While further trials are needed to validate safety, durability, and scalability, this innovation provides a strong foundation for future immuno-compatible regenerative treatments in diabetes.
For enquiries info@jothydev.net.
Please visit: jothydev.net | research.jothydev.com | diabscreenkerala.net | jothydev.com/newsletter




