 |
||||
|
||||
| 6. Diabetes Medicine Updates | ||||
Lucentis approved for treating diabetic macular edema |
||||
| The FDA has approved Lucentis to treat diabetic macular edema, a common, vision-threatening complication of diabetes. Lucentis is given via a monthly injection. In diabetic macular edema, fluid leaks into the macula (the middle of the retina), which is responsible for providing crisp, clear vision. The fluid swells the macula and blurs vision. Lucentis is already approved to treat other eye disorders, including wet age-related macular degeneration and macular edema caused by blockage of the veins that carry blood away from the retina. | ||||
| Read More | ||||
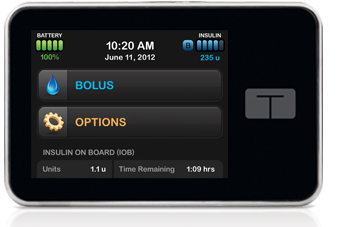
| Tandem T:slim Touchscreen Insulin Pump | ||||
Tandem Diabetes Care™ t:slim™ Insulin Delivery System is the first insulin pump approved by the FDA with a touch screen, that embodies a fresh new design approach and offers the most requested features asked for in a next-generation pump. The t:slim™ Insulin Delivery System with its vivid color touch screen is currently the smallest FDA-cleared insulin delivery system and is up to to 25% slimmer than other 300 unit pumps. It’s recharged using a standard micro-USB cable. It has a revolutionary pump system powered by pressure with a micro dosing mechanism that can deliver doses as small as 0.001 u. Unlike cartridge pumps that are connected directly to an infusion line, the t:slim has no motor and far fewer parts than current insulin pumps. It will also not over deliver insulin (excess delivery can be as large as 1.0 to 1.4 units) during the pressure drops that occur in flight as the pressure drops inside an airplane after takeoff. |
 |
|||
| Read more | ||||